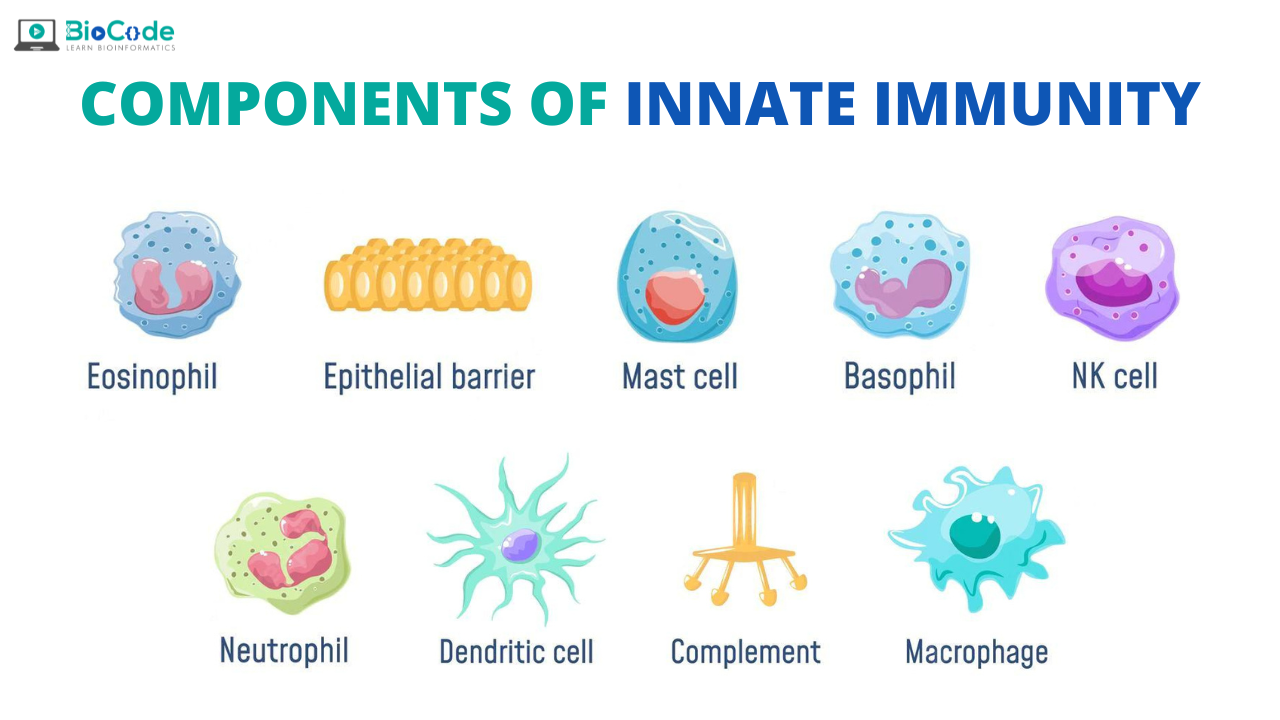
The components of the innate immune system includes: Epithelial cells and Sentinel cells in tissues (macrophages, dendritic cells, mast cells, and others), Innate lymphoid cells, including NK cells and several plasma proteins.
Epithelial Barriers
The major interfaces between the body and the external environment is the skin, gastrointestinal tract, respiratory tract, and genitourinary tract. These are protected by continuous epithelia that provide physical and chemical barriers against infection. Keratin on the surface of the skin and mucus secreted by mucosal epithelial cells prevent microbes from encountering and infecting the epithelia. Epithelial cells also produce peptide antibiotics, called defensins and cathelicidins, which kill bacteria and thus provide a chemical barrier against infection. Epithelia contain lymphocytes called intraepithelial T lymphocytes that belong to the T cell lineage but express antigen receptors of limited diversity.
Functions of Epithelia in Innate Immunity:
Epithelia present at the portals of entry of microbes provide physical barriers formed by keratin (in the skin) or secreted mucus (in the gastrointestinal, bronchopulmonary and genitourinary systems) and by tight junctions between epithelial cells. Epithelia also produce antimicrobial substances (e.g., defensins) and harbor lymphocytes that kill microbes and infected cells.
Phagocytes: Neutrophils and Monocytes/ Macrophages Neutrophils:
The two types of circulating phagocytes, neutrophils and monocytes, are blood cells that are recruited to sites of infection. They recognize and ingest microbes for intracellular killing. Neutrophils, also called polymorphonuclear leukocytes (PMNs), are the most abundant leukocytes in the blood, numbering 4000 to 10,000 per µL. The production of neutrophils is stimulated by cytokines which are known as colony-stimulating factors (CSFs). They are secreted by many cell types in response to infections. Neutrophils are the first cell type to respond to most infections, particularly bacterial and fungal infections. Neutrophils ingest microbes in the circulation, and they rapidly enter extravascular tissues at sites of infection, where they also phagocytose and destroy microbes. Neutrophils express receptors for products of complement activation and for antibodies that coat microbes. Neutrophils live for only a few hours in tissues. They are the early responders.
Monocytes
They also ingest microbes in the blood and in tissues. During inflammatory reactions, monocytes enter extravascular tissues and differentiate into cells called macrophages. Unlike neutrophils, they survive in these sites for long periods. Blood monocytes and tissue macrophages are two stages of the same cell lineage (mono nuclear phagocyte system). Macrophages are also found in all connective tissues and organs of the body.
Macrophages:
Macrophages serve several important roles in host defense: they produce cytokines that induce and regulate inflammation. They ingest and destroy microbes, and they clear dead tissues and initiate the process of tissue repair. Pattern recognition receptors, including TLRs and NLRs, recognize products of microbes and damaged cells and activate the macrophages. Phagocytosis is mediated by cell surface receptors. Some of these phagocytic receptors activate the microbial killing functions of macrophages.
Macrophage Activation:
Macrophages may be activated by two different pathways.
⦁ Classical macrophage activation
⦁ Alternative macrophage activation
Classical macrophage activation:
It is induced by innate immune signals, such as from TLRs, and by the cytokine IFN-γ, which may be produced in both innate and adaptive immune responses. Classically activated macrophages, also called M1, are involved in destroying microbes and in triggering inflammation.
Alternative macrophage activation:
It occurs in the absence of strong TLR signals and is induced by the cytokines IL-4 and IL-13; these macrophages, called M2, appear to be more important for tissue repair and to terminate inflammation.
Dendritic Cells
Dendritic cells respond to microbes by producing numerous cytokines that serve two main functions: they initiate inflammation, and they stimulate adaptive immune responses. By sensing microbes and interacting with lymphocytes, especially T cells, dendritic cells constitute an important bridge between innate and adaptive immunity.
Mast Cells
Mast cells are bone marrow–derived cells with abundant cytoplasmic granules that are present in the skin and mucosal epithelium. Mast cells can be activated by microbial products binding to TLRs. Mast cell granules contain vasoactive amines such as histamine that cause vasodilation and increased capillary permeability. It also produces proteolytic enzymes that can kill bacteria or inactivate microbial toxins. Mast cell products provide defense against helminths and other pathogens and are responsible for symptoms of allergic diseases.
Innate Lymphoid Cells
Innate lymphoid cells (ILCs) are lymphocyte-like cells that produce cytokines. They perform functions like those of T lymphocytes but do not express T cell antigen receptors (TCRs). ILCs have been divided into three major groups based on their secreted cytokines. These groups correspond to the Th1, Th2, and Th17 subsets of CD4+ T cells. ILCs provide early defense against infections and guide the subsequent T cell response.
Natural Killer Cells
Natural killer (NK) cells recognize infected and stressed cells and respond by killing these cells and by secreting the macrophage activating cytokine IFN-γ. NK cells contain abundant cytoplasmic granules and express some unique surface proteins but do not express immunoglobulins or T cell receptors. On activation by infected cells, NK cells empty the contents of their cytoplasmic granules into the extracellular space at the point of contact with the infected cell. The granule proteins then enter infected cells and activate enzymes that induce apoptosis. With CTLs, NK cells function to eliminate cellular reservoirs of infection and eradicate infections by obligate intracellular microbes, such as viruses.
Functions of natural killer (NK) cells:
NK cells kill host cells infected by intracellular microbes, thus eliminating reservoirs of infection.
NK cells respond to interleukin-12 (IL-12) produced by macrophages and secrete interferon-γ (IFN-γ), which activates the macrophages to kill phagocytosed microbes.
Activating And Inhibitory Receptors Of Natural Killer (NK) Cells:
Healthy host cells express self class I major histocompatibility complex (MHC) molecules, which are recognized by inhibitory receptors, thus ensuring that NK cells do not attack normal host cells. Note that healthy cells may express ligands for activating receptors or may not express such ligands, but they are not attacked by NK cells because they engage the inhibitory receptors.
NK cells are also activated by infected cells in which ligands for activating receptors are expressed (often at high levels) and class I MHC expression is reduced so that the inhibitory receptors are not engaged. The result is that the infected cells are killed.
Complement System
The complement system is a collection of circulating and membrane-associated proteins that are important in defense against microbes. Many complement proteins are proteolytic enzymes, and complement activation involves the sequential activation of these enzymes. The complement cascade may be activated by any of three pathways.
The Alternative Pathway
The alternative pathway is triggered when some complement proteins are activated on microbial surfaces and cannot be controlled, because complement regulatory proteins are not present on microbes (but are present on host cells). The alternative pathway is a component of innate immunity.
The classical pathway is most often triggered by antibodies that bind to microbes or other antigens and is thus a component of the humoral arm of adaptive immunity.
The lectin pathway is activated when a carbohydrate-binding plasma protein, mannose-binding lectin (MBL), binds to terminal mannose residues on the surface glycoproteins of microbes. This lectin activates proteins of the classical pathway, but because it is initiated by a microbial product in the absence of antibody.
Pathways Of Complement Activation:
The activation of the complement system (the early steps) may be initiated by three distinct pathways, all of which lead to the production of C3b. C3b initiates the late steps of complement activation, culminating in the formation of a multiprotein complex called the membrane attack complex (MAC), which is a transmembrane channel composed of polymerized C9 molecules that causes lysis of thin-walled microbes. Peptide by-products released during complement activation are the inflammation inducing C3a and C5a.
Functions Of Complement System
The complement system serves three main functions in host defense.
Opsonization and phagocytosis:
C3b coats microbes and promotes the binding of these microbes to phagocytes.This process of coating a microbe with molecules that are recognized by receptors on phagocytes is called opsonization.
Inflammation:
Some proteolytic fragments of complement proteins, especially C5a and C3a, are chemo attractants for leukocytes (mainly neutrophils and monocytes), so they promote leukocyte recruitment (inflammation) at the site of complement activation.
Cell lysis:
Complement activation helps in the formation of a polymeric protein complex that inserts into the microbial cell membrane which disturbs the permeability barrier and causing either osmotic lysis or apoptosis of the microbe.
Cytokines of Innate Immunity
Cytokines are soluble proteins that mediate immune and inflammatory reactions and are responsible for communications between leukocytes and between leukocytes and other cells. Most of the molecularly defined cytokines are called interleukins. In response to microbes, dendritic cells, macrophages, mast cells, and other cells secrete cytokines that mediate many of the cellular reactions of innate immunity.