Drugs interact with your body in a number of ways, depending on how you take the drug and where the drug may act in your body. After you take a drug, your body needs to break it down and get it to the intended area. Your DNA can affect multiple steps in this process to influence how you respond to the drug.
Pharmacogenomics
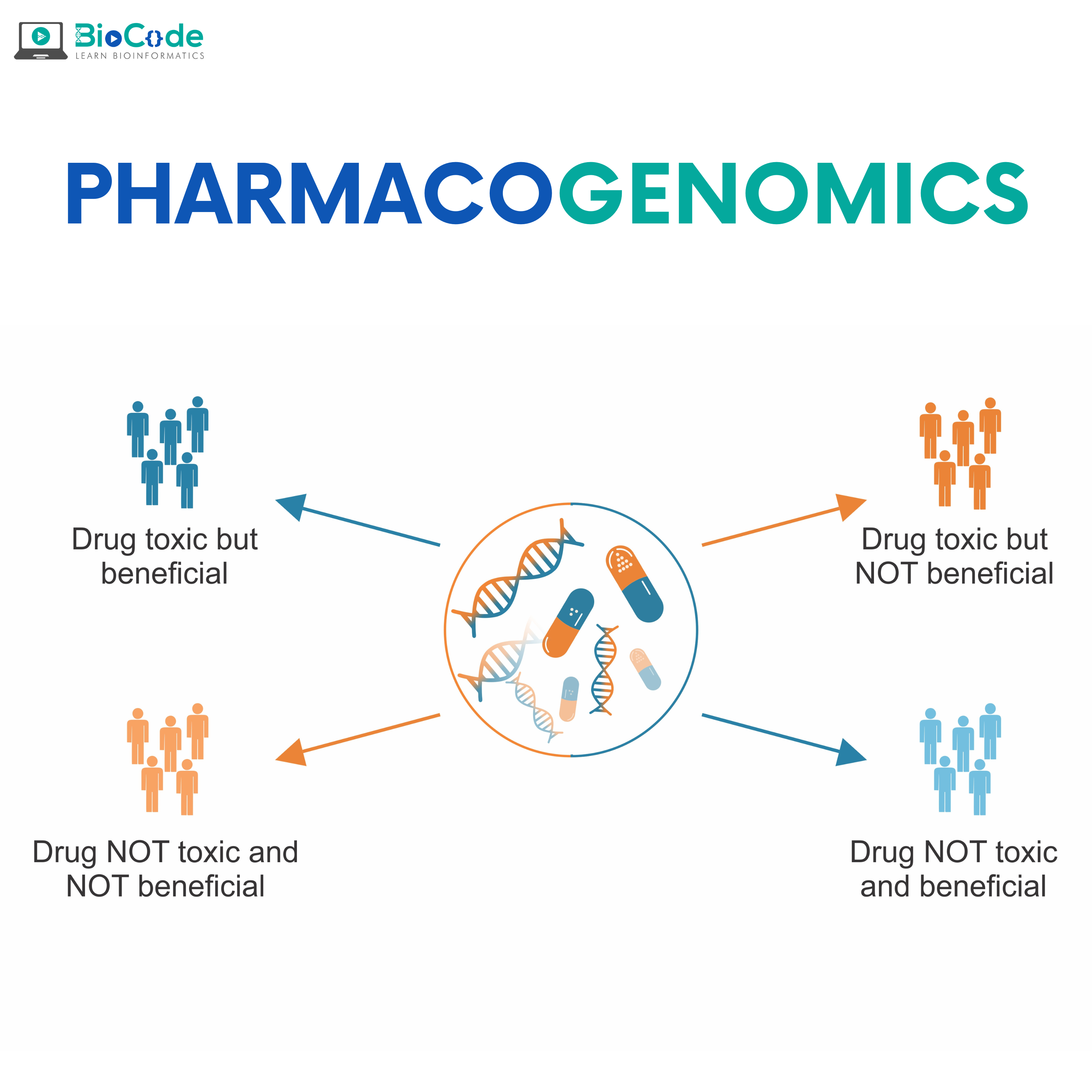
Pharmacogenomics is the study of how genetic variants influence drug efficacy and toxicity. This field combines pharmacology (the science of drugs) and genomics (the study of genome, genes, and their functions) to develop efficient, and safe medications that can be prescribed based on a person’s genetic makeup.
Pharmacogenomics inspects how your DNA is affected by the way you respond to drugs. The long-term goal of pharmacogenomics is to help doctors select the drugs that are best suited for each person. It is part of the field of precision medicine, which aims to treat each patient individually.
Cancer Pharmacogenomics
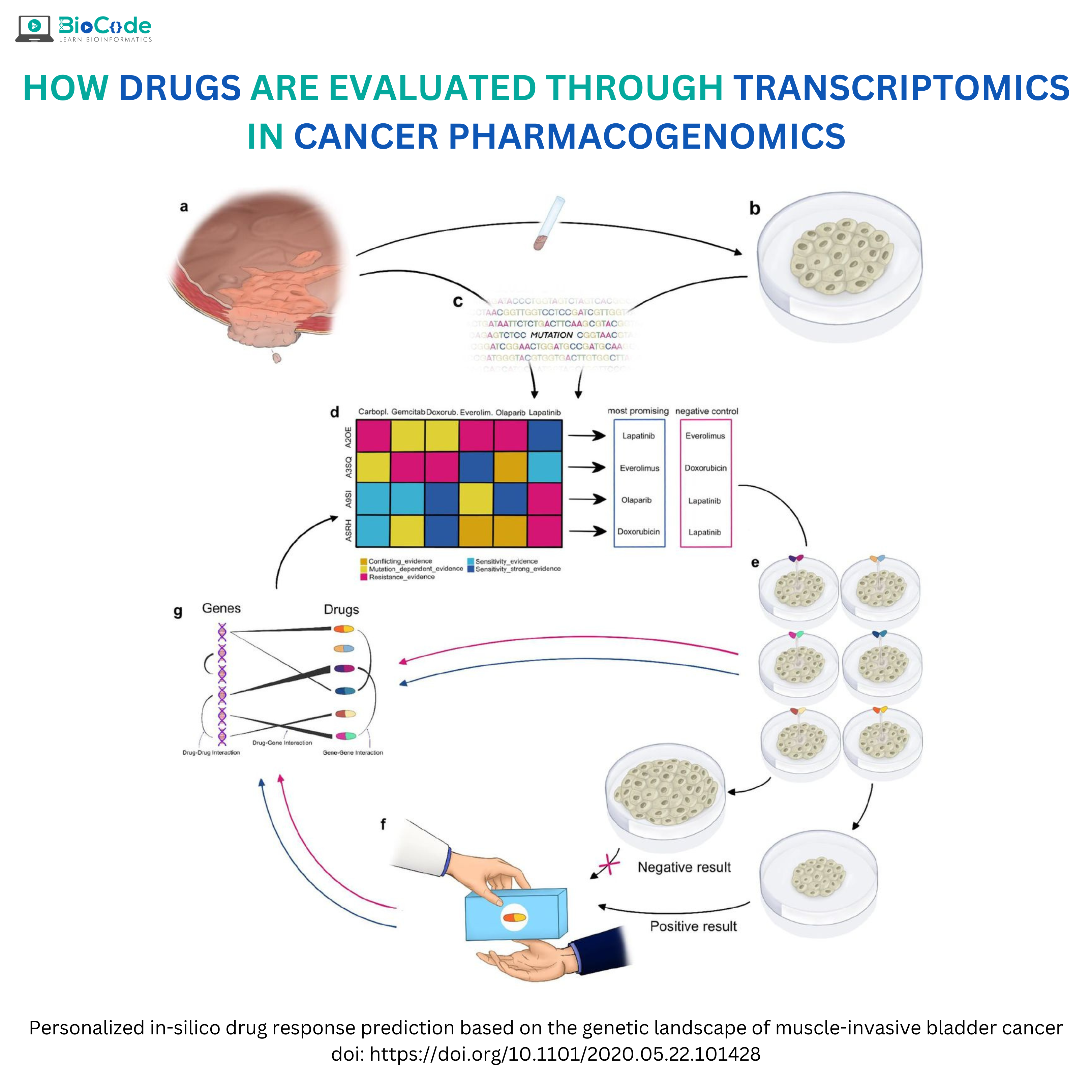
Cancer is among the leading causes of death worldwide. It is a disease that causes cells to divide uncontrollably and spread into surrounding tissues. There are many types of cancer treatments available which depend on the type of cancer you have and how advanced it is. The sensitivity to cancer therapy varies significantly among patients. The important factor to counter here is how the patient’s body will react to each treatment. The ability to predict how a cancer patient will respond to a particular treatment is the goal of personalized oncology.

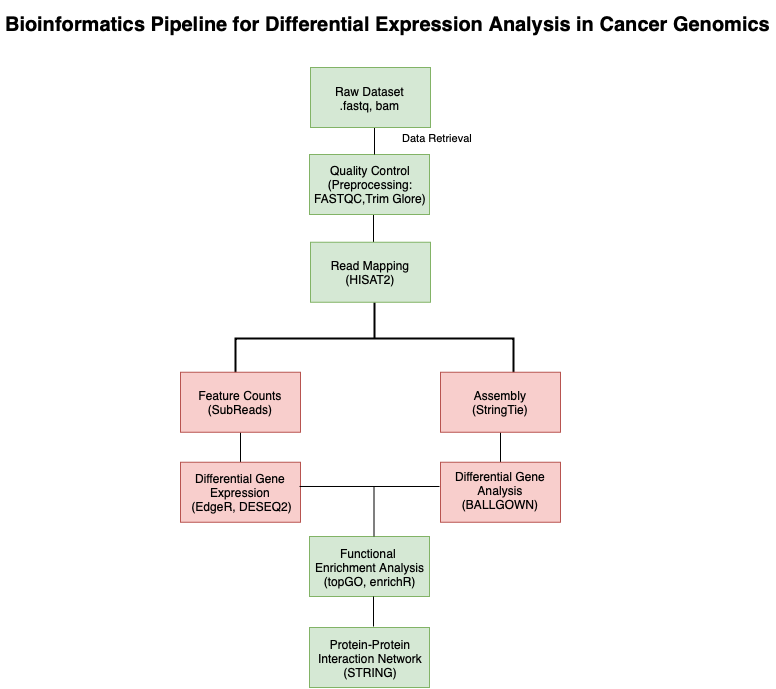
RNA-Sequencing in Cancer Pharmacogenomics
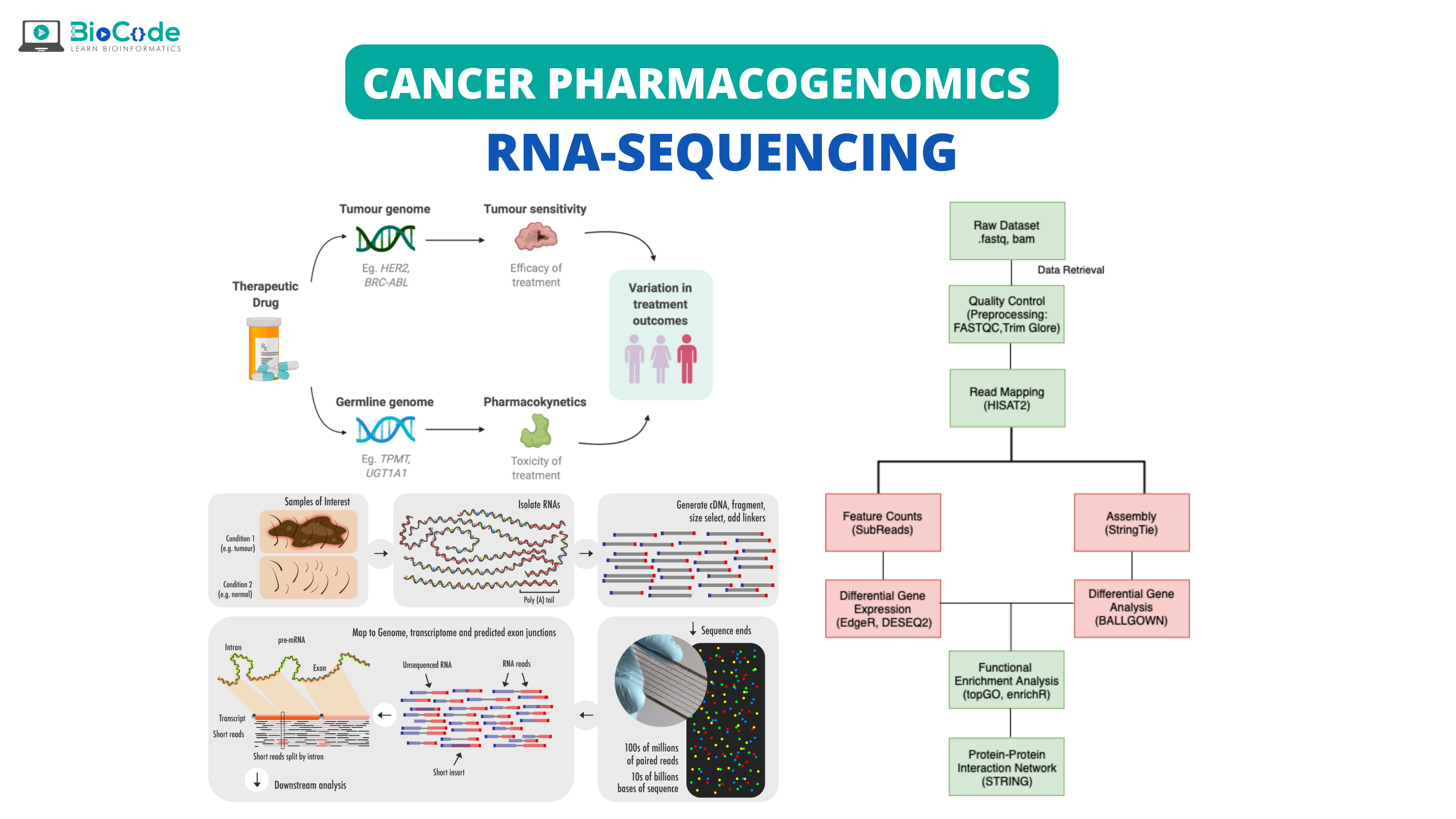
With the advent of RNA sequencing over a decade ago, diagnosis and identification of cancer entered a new phase that enabled more accurate analysis that is difficult to approach and analyze. RNA-seq is being widely used for the research on gene expression in various malignancies and has been found as an effective tool in the exploration of carcinogenesis, identification of tumor biomarkers, and development of new therapeutic drugs and strategies.
Analysis of gene expression allows researchers to explore the molecular basis of cancer and provide information on tumor progression. Various assays based on gene expression analysis have been developed to guide personalized therapeutic pathways in oncology. RNA-Seq has enabled insight into the molecular mechanisms underlying cancer which has given rise to numerous trials related to transcriptome-based personalized oncology.
The expanding availability of transcriptome profiling has enabled the introduction of transcriptomics in clinics. Several prognostic and predictive gene expression analysis, as well as cancer biomarkers, have been utilized in clinical oncology. The change in the standard cancer classification from histopathological and clinical to molecular has opened a more customized perspective for tumor diagnostics and therapy. With the help of cancer pharmacogenomics, it has become possible to predict how a certain drug will function in the diseased body.
Examples of Cancer Pharmacogenomics
Advancements have been made in breast cancer research after performing gene expression profiling to establish molecular classification and identification of therapeutic/diagnostic targets in breast cancer. Other than this various efforts were made in colorectal cancer (CRC) as well. In this research for colorectal cancer, four consensus molecular subtypes of CRC were distinguished. Similarly, another study uncovered differences in origin of ovarian cancer.
Learn Cancer Genomics: NGS (Whole Exome Sequencing) Variant Calling Using Linux
An example of gene expression profiling in the selection of cancer biomarkers can be found in gastrointestinal tract tumors. Many RNAs were found to be useful in precision oncology for instance miR-106b, miR-20a, and miR-221 were confirmed as early detection biomarkers for gastric cancer. The piRNAs, which mediate transcriptional and post-transcriptional gene silencing machineries, may serve as biomarkers for early diagnosis, treatment and prognosis of renal cancer, hepatocellular carcinoma, glioblastoma, and gastric cancer.